A brick of fired earth holds more than shape and decoration. It locks in traces of the hillside where the clay was dug, records the temper added by a potter’s hand, and preserves the heating history of a single firing chamber. Those traces are not metaphors. They are minerals, glass, and pores that carry chemical and textural signals strong enough to identify a workshop, a clay source, sometimes even a particular kiln load. When archaeologists say a pot’s fabric is local or that a sherd “belongs to La Graufesenque,” they are not guessing. They are reading the kiln’s fingerprint.
This essay explores how minerals name a kiln. It introduces the core methods that let researchers connect a pot to a place: hand specimen fabric analysis, thin section petrography, X-ray and neutron methods, electron microscopy, and isotopes. It explains what those methods see in real objects: quartz and grog in a cooking pot, iron oxides in a red gloss slip, carbonates that betray a lower firing, volcanic glass that points to specific valleys. It walks through field indicators like wasters and kiln furniture, then ties them to laboratory data. Case studies from Roman terra sigillata, Dressel 20 amphorae, and medieval wares show how stamps, shapes, and mineral chemistry fit together. The result is a practical guide to the science behind phrases like fabric, provenance, and workshop.

What a Pot Records When It Meets Fire
Every ceramic begins as a mixture of clay minerals, water, and nonplastic inclusions. Clays are microscopic sheet silicates like kaolinite, illite, and smectite. They respond to water by swelling or remaining stable, and they respond to heat by transforming. Potters rarely rely on clay alone. They add temper, a deliberately chosen coarse component that changes how a vessel dries, shrinks, and resists thermal shock. Temper can be crushed pottery, called grog, or fragments of quartz, feldspar, crushed limestone, shell, or volcanic sand.
When the vessel enters a kiln, most clays dehydrate and begin to reorganize. Around 573 degrees Celsius quartz passes through a reversible alpha to beta transition that leaves a detectable texture in the final fabric. Between about 600 and 800 degrees Celsius, carbonates like calcite and dolomite decompose. Around 900 to 1050 degrees Celsius, many common clays begin to vitrify, forming glass that surrounds the remaining grains. Iron reads firing conditions like a thermometer. Hematite gives red hues in oxidizing firings, magnetite forms under more reducing conditions. A bright red gloss slip is often a colloidal clay layer rich in iron that sinters into a dense, thin film studded with submicroscopic crystals.
Those transformations lock a sequence of decisions into the pot: which clay pit, which temper, how the paste was mixed, what atmosphere the kiln breathed, what temperature it reached, how long it soaked. Because geology varies from valley to valley, and because workshop practice varies from crew to crew, the combination can be distinctive. That distinctiveness is what archaeologists measure when they “name” a kiln.

The Vocabulary of Fabric and Provenance
Archaeologists use two similar words with care. Provenience names the findspot, the exact place a sherd was excavated. Provenance names the origin, the place and context in which an object was made. Fabric sits at the heart of provenance. Fabric refers to the visible and microscopic makeup of a ceramic body: clay matrix, temper grains, voids, and any added slip. A fabric series is a set of recurring recipes within a production tradition.
In practice, a researcher starts with the body. A hand lens reveals grain size, inclusions, and voids. A fresh break shows the fracture: conchoidal in dense, well-vitrified ware, laminar or platy in micaceous fabrics. Color suggests firing atmosphere. A quick dip of dilute hydrochloric acid reveals carbonates by fizzing. A magnet can pick up iron-rich particles or magnetic kiln ash. These low-tech steps matter, because they guide sampling and sometimes solve the problem outright. A coarse, quartz-rich cooking pot with grog and soot-blackened exteriors points toward utilitarian table or kitchen wares, not fine imported slipware.
The next step is thin section petrography. A slice of the body is ground to about 30 micrometers thick, mounted on glass, then viewed under a polarizing microscope. In cross-polarized light, feldspars flash twinning, quartz shows undulatory extinction, volcanic glass shows brown or colorless patches, grog reveals angular grains of old ceramic. The spatial relationships matter: whether inclusions are sorted or bimodal, whether voids are elongated by wheel forming or uneven by hand building, whether mica flakes lie in preferred orientations. Many workshops are defined by a particular mix of inclusions, like limestone microfossils in calcareous clay or basaltic sand in a volcanic province. That recipe can be as diagnostic as a stamp.

The Toolkit: From Light to Neutrons
Petrography and optical methods
The thin section remains the backbone of ceramic sourcing because it links directly to geology and craft practice. Petrography identifies inclusions, discriminates between natural aplastics and added temper, and recognizes recycled grog. It reads forming traces, like aligned mica from wheel throwing. It estimates firing by the onset of vitrification. It distinguishes fabrics that can look identical to the eye.
Complementary optical methods include reflected light for opaque phases and Raman spectroscopy for identifying iron oxides, carbon, or specific pigments in slip layers. In chalky calcareous fabrics, simple cold hydrochloric acid tests paired with microscopic observation can distinguish sparry calcite recrystallized after firing from remnant shell.
X-ray fluorescence and diffraction
X-ray fluorescence, or XRF, measures the concentrations of elements from sodium through uranium in a sample’s surface. Bench-top XRF devices measure pressed pellets or fused glass beads and provide repeatable major and trace element data. Portable XRF, or pXRF, places a handheld analyzer on the sherd surface. It is fast and nondestructive, which is vital in museums and on sites, but it demands careful calibration and an understanding of biases. Glaze and slip can differ sharply from the body. Weathering can leach alkalis. Surface roughness and curvature can skew readings. Even with those caveats, pXRF remains a powerful way to separate groups, test hypotheses about trade, and pre-screen samples before destructive methods.
X-ray diffraction, or XRD, identifies crystalline phases. It answers questions that chemical totals cannot. Is the red slip hematite or iron-rich glass with nanocrystals. Did calcite survive firing or recrystallize during burial. Is the clay matrix dominated by illite or kaolinite. In early or low-fired wares, XRD can still see clay minerals that would be destroyed at higher temperatures, which helps estimate firing regimes.
Neutron activation analysis and ICP methods
Instrumental neutron activation analysis, or NAA, became a workhorse for provenance in the late twentieth century and remains essential where precision matters. A powdered sample is irradiated in a reactor. Neutron capture produces radioactive isotopes that decay with element-specific gamma emissions. The resulting trace element patterns, especially for rare earths, provide fingerprints that can distinguish clays from neighboring valleys. Because the method analyzes the bulk, it is less sensitive to surface contamination than XRF. The price is destructive sampling and the need for specialized facilities.
Inductively coupled plasma methods add flexibility. ICP-OES measures many major and minor elements. ICP-MS pushes deep into trace elements. When paired with laser ablation, LA-ICP-MS can target thin slips or micro-domains. These methods generate multivariate data that cluster nicely in principal component or discriminant analysis, which lets researchers match unknown sherds to reference groups with formal statistics.
Electron microscopy and microanalysis
Scanning electron microscopy, SEM, magnifies textures and interfaces. Backscattered electron imaging highlights density differences, so glassy matrix and crystalline inclusions pop apart. Energy-dispersive spectroscopy, EDS, reads elemental composition spot by spot. This combination reveals vitrification gradients across body to slip, identifies crystals in glossy red slips as hematite or spinel, and catches reaction rims around calcite grains where lime interacted with a silicate melt. It is also excellent for failure analysis in wasters, showing bloating from trapped gases, warped supports, or sintered bridges between stacked vessels.
Isotopes and archaeomagnetism
Isotope systems add another layer for specific questions. Strontium isotopes often track local geology and can separate marine shell temper from freshwater carbonate. Lead isotopes can trace lead-bearing glazes or pigments. Oxygen isotopes in carbonates can sometimes preserve signatures of water sources, although firing and diagenesis complicate interpretation. Archaeomagnetism reads the alignment of magnetic minerals set during cooling to estimate the last cooling direction or date when appropriate reference curves exist. Thermoluminescence and optically stimulated luminescence date last firing or last sun exposure, valuable for authenticity tests.
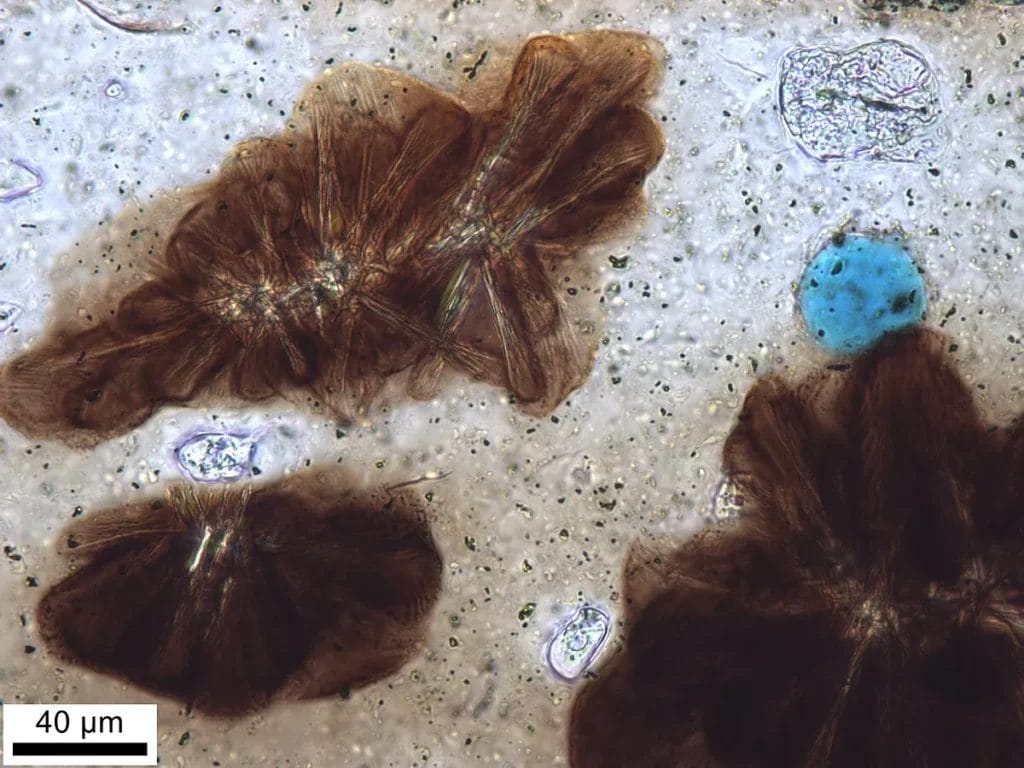
Field Clues That Narrow the Search
Laboratory signatures only work if the fieldwork sets them up. Some features on a site immediately suggest a kiln nearby.
Wasters. A waster is a misfired pot discarded at or near the kiln because it is warped, fused to a neighbor, underfired, or bloated. A concentration of wasters is a production signature. If they include the same fabric as later sherds on a settlement, the case for local manufacture strengthens.
Kiln furniture. Spacers, perforated floors, saggars, and columns leave distinctive ceramic debris. Perforated floors from up-draft kilns look like thick slabs with regular vents. Bars and rings carry glaze drips and ash, which can be analyzed to reconstruct firing atmospheres.
Clay pits and quarries. Exposed clayey beds, old extraction scars, or pits with slickensides point toward raw material. In many regions clay sources sit within a few kilometers of workshops to reduce transport costs, although large production centers may draw from multiple pits to stabilize supply.
Stamped handles and mold-made relief. Stamps on amphora handles and maker’s marks on fine wares tie a sherd to a workshop name. When those marks co-occur with a diagnostic fabric, the resulting reference group stabilizes attribution for unmarked sherds.

Case Study: Red Gloss Tables Ware and the Gaulish Kilns
Terra sigillata, the glossy red tableware of the Roman world, demonstrates how minerals, stamps, and kilns align. The production quarter at La Graufesenque, near modern Millau in southern France, operated on an industrial scale in the first and second centuries CE. Excavation revealed huge kilns, including a monumental structure capable of firing tens of thousands of vessels in a single cycle. Firings were organized into stories with perforated floors and hollow supports that channeled heat. Lists of potters and counts sometimes fired with the cargo survive on fragments, a rare documentary window onto a kiln load.
Fabrics and slips from La Graufesenque are distinctive. Petrography often shows fine, well-sorted inclusions with sparse mica. Many bodies are relatively calcareous compared to some central Gallic wares, a difference that shows up in both thin sections and bulk chemistry. The red slip is a sintered iron-rich coating that can display tiny crystals embedded in glass when viewed under SEM. When chemical data from bodies and slips are plotted, La Graufesenque forms tight clusters that separate from neighboring centers like Lezoux and central Italian Arezzo. The separation comes from both geology and practice: clay choices, levigation methods, temper, and firing recipes differ even when forms and decorative motifs look similar.
Sampling along a road or a wall line shows how these clusters help with trade. A fort on the northern frontier can yield a spectrum of red gloss sherds. Some carry stamps from southern Gaul, others from central Gaul, a few from Italian workshops. Their bodies and slips divide into clusters that match reference groups, so even abraded, unstamped fragments can be assigned with confidence. If a layer dates to a specific decade, the relative shares of workshops date the flow of goods as markets shift.

Case Study: Dressel 20 Amphorae and Monte Testaccio
Dressel 20 amphorae are the globular oil containers shipped from Baetica, modern Andalusia, to Rome and elsewhere. Their thick handles and shoulder stamps provide names. Their fabrics contain coarse quartz and often calcareous components that are easy to spot in hand specimen. Chemical analyses show that Baetican clays form tight compositional groups distinct from Gallic or African centers. The mound of broken amphorae at Monte Testaccio in Rome formed from the systematic dumping of these oil containers. Painted tituli picti record the weight of oil and officials who measured it. Stamps and paint provide one kind of label. Trace element patterns in the bodies provide another. Together they allow archaeologists to see how specific estates or workshops fed Rome in different periods, and how that supply changed across the second and third centuries.
How Much Clay Do You Need to Name a Kiln
Sampling is a balance between information and preservation. A responsible program starts with non-destructive steps, then uses minimal destructive sampling targeted to answer specific questions.
A common workflow looks like this:
- Define the question. Are we separating imported from local ware in one site. Are we matching a museum fragment to a workshop. Are we testing whether two fabrics thought to be the same are actually distinct.
- Build a reference set. Include wasters, kiln furniture, and stamped pieces from known workshops whenever possible. Add raw clays from suspected source pits. Without a reference set, assigning unknowns becomes speculation.
- Group visually. Record fabric traits, temper type, and macroscopic color using standardized charts. Create preliminary groups that can be tested later.
- Test with quick tools. Use pXRF to see whether obvious groups emerge in elemental data. Scan body and slip separately. Reject or adjust groupings that do not make chemical sense.
- Select samples for thin section and bulk analysis. Pick a small number from each group for petrography and either XRF on pellets, ICP, or NAA. Avoid sherds with heavy burial alteration.
- Analyze statistically. Principal component analysis or discriminant functions help visualize clusters. The goal is not a pretty plot, it is a stable group definition that you can apply to unknowns.
- Cross-check with geology. Thin section minerals must make sense for the claimed source. If a supposed workshop sits on chalk but the fabric is volcanic, the story is wrong.
- Publish the criteria. Describe inclusions, matrix, and chemistry clearly enough that others can replicate your assignments. Include images and data tables. A kiln’s fingerprint only works if multiple teams can read it.

What Minerals Say About Craft, Not Just Place
Clay recipes are technological choices. Grog temper, recycled from broken pots, makes a body thicken and resist thermal shock, which suits cooking vessels. Quartz temper rises in abundance in vessels that need to withstand rapid heating. Carbonate temper works at lower temperatures but fails if fired too high, since lime particles expand and can cause spalling. Volcanic sand adds high-temperature stability and leaves heavy minerals like pyroxene and amphibole in the fabric. Texture shows forming methods. Wheel throwing aligns platy mica and elongates pores around 30 to 45 degrees to the rim. Hand building leaves more random pore patterns. Burnishing closes surface pores and leaves a smooth boundary between slip and body.
Red slips deserve special attention, because they carry some of the clearest technological signals. Many red gloss slips start as levigated clays, enriched in fine iron-bearing particles. The firing regime must pass through the right temperature range long enough to sinter the slip without blistering. The resulting microstructure often shows fine crystals embedded in a glassy matrix. Under SEM these crystals can be identified as hematite or related phases. Their size and density vary by workshop, and those differences can sometimes be used to separate centers that copy each other’s forms.
The same logic extends to glaze and paint. A lead glaze carries lead isotope signatures that can, in favorable cases, point to a particular ore district. Egyptian blue pigment inlays or paints carry a distinct copper-rich silicate with a crystalline lattice easily seen in SEM. Each of these material choices narrows origin and reveals knowledge systems within workshops.
The Limits of Fingerprints: Mixing, Markets, and Movement
Mineral fingerprints are powerful, but they are not magic formulae. Clay beds can be broad and variable, and potters often mix clays deliberately to improve workability. Workshops move through time. A production group active in one century may adopt new sources or recipes in the next. Clay sources change with ownership or access to land. Migrant potters carry recipes across regions and teach apprentices who adapt them to local resources. Traded pots collect contaminations on their surfaces from soil, salts, and handling.
Analytical methods carry their own limits. pXRF reads only the top micrometers, so dust, slips, and patina can dominate the result. NAA and ICP require grinding a piece of the object, which museums often cannot permit for complete vessels. Thin sections destroy a slice and require specialized skill to interpret consistently between labs. Isotope systems can be reset by weathering or may overlap between nearby districts. Statistical separations can be crisp in one dataset and blurry in another if sample selection changes.
These limits do not refute kiln fingerprinting. They explain why multiple methods together, plus well-documented reference sets from production centers, produce the most reliable attributions. A stamp on an amphora handle and a calcareous, quartz-rich body cluster make a strong Baetican case. A waster dump near a kiln and a petrographic match tie a fabric to a workshop. A distinctive slip microstructure and a rare earth element pattern from NAA separate two look-alike red gloss horizons.
How Workshops Scale: The Industrial Archaeology of Firing
Naming a kiln also means understanding how kilns were organized. Small workshops serve a town or a rural district. They build clamp kilns or modest up-draft structures. Their wasters cluster in modest dumps. Their fabrics stay close to one clay bank, sometimes with seasonal variability as pits flood or dry. Large workshops operate like factories. They stabilize clay supply by drawing from multiple pits and blending. They invest in massive kilns, multiple firing floors, and permanent ventilation. Their waster dumps grow into hills. Their output carries dozens of potter’s names and stylistic hands. Their distribution can be tracked across provinces.
La Graufesenque exemplifies the large end. Another model sits in central Italy at Arezzo and its satellite centers, where early experiments with red gloss slip matured into an export industry. In both cases, mineral fingerprints tell stories of scale. Large, stable clusters in body chemistry suggest controlled raw materials and disciplined recipes. Minor shifts in slip chemistry through time show workshop adjustments. The spatial distribution of fabrics on distant sites shows market reach and competition. By reading minerals, archaeologists reconstruct not just where a pot was fired, but how a production system flexed under demand.
Beyond the Mediterranean: Universals in Clay
The logic of kiln fingerprinting is not confined to Romans. Neolithic Linear Pottery Culture vessels in central Europe can be grouped by petrography into village-scale fabrics. Mesoamerican wares have been assigned to workshops by pXRF and NAA, with volcanic tempers pointing to specific river valleys. Early Islamic glazed wares show lead and tin signatures that track ore districts and trade. Medieval European cooking pots with shell temper reveal freshwater versus marine shell in thin section and by strontium isotopes. In each case, geological diversity and craft choice produce diagnostic combinations.
The approach travels to crucibles and molds as well. Metallurgical ceramics often carry residual lead, copper, or arsenic from their contents. pXRF can map those residues and, combined with fabric, identify workshop zones dedicated to metalworking on multi-craft sites. Kiln furniture can be sampled as a proxy for firing atmospheres and fuel types, since ash and fluxes soak into supports during operation.
Practical Advice for Readers on Site or in a Gallery
If you handle sherds in the field, a few habits improve your sense for fabrics.
Start with the break. Fresh breaks show inclusions clearly. Use the smallest possible fragment to avoid damage. Keep a hand lens and note the size range of inclusions. Are they well sorted or mixed. Scratch the surface with a steel pin to feel hardness and to test the difference between compact slip and chalky body. Drop a tiny bit of dilute acid on a hidden spot to test for carbonates. Note whether the surface was wiped, burnished, or left rough. Touch the rim to see whether it is rounded by abrasion or sharp from a fresh break.
Train your eye with reference pieces. Museums sometimes display labeled sherd sets with known fabrics. Photographs rarely capture subtle pore shapes or mica glints as well as direct light. Compare your field sherds to the reference textures and bring those comparisons into the lab as explicit hypotheses. Rather than saying “local,” say “quartz-rich sandy fabric with mica, resembling known group X.”
Finally, keep context. A kiln fingerprint is only one line of evidence. Stratigraphy, associated finds, and radiocarbon dates frame what a fabric identification means. A nonlocal amphora sherd in a local cooking pot dump tells a different story than a yard littered with identical wasters.









